Genus/species (former genus/species): Hansenula anomala (now merged into Pichia anomala; synonyms with Willia anomala, Endomyces anomalus, Wickerhamomyces anomalus)
Classification: Ascomycete
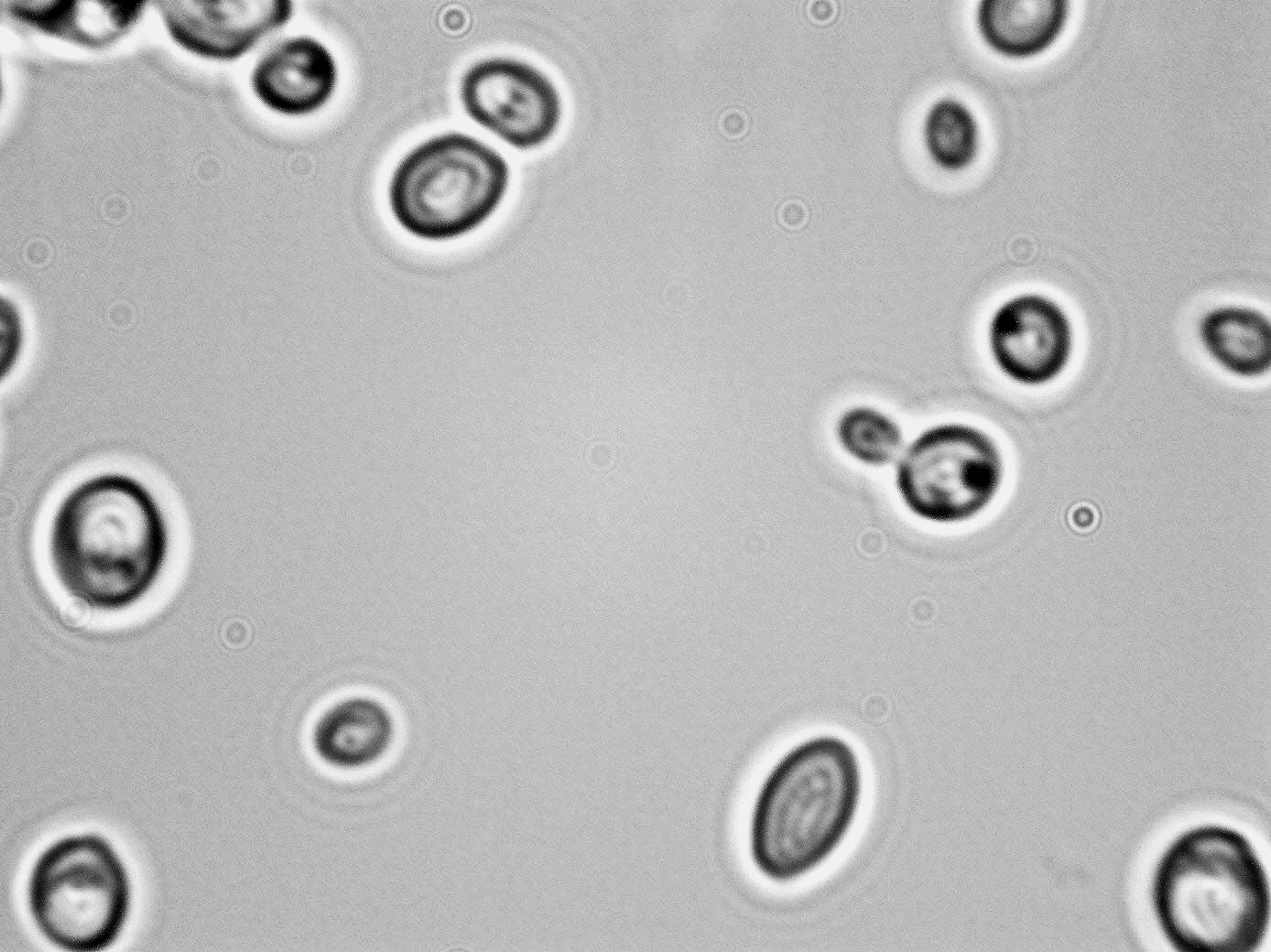
Morphology:
- Cell: Spheroidal to elongate (1.9-4.1)X(2.1-6.1)µm singly, pairs, small clusters; ovoid, ellipsoidal, or cylindrical, multilateral budding. No true hyphae. Pseudohyphae form as chains of ovoid or cylindroidal cells.
- Colony: Malt agar: Butyrous, tan in colorWL: cream-color, flat, smooth, creamy
- Spore: Usually heterothallic, but sporogenous diploid type form one to four hat-shaped ascospores
- Zygote: Diploid cells convert directly to asci.
- Ascus: When formed, contains one to four hat-shaped spores, converted directly from sporogenous cells.
- Liquid Growth: Pellicle, varies from thin/smooth to thick/folded.
 |
 |
 |
 |
Physiological Traits:
Ferment glucose, sucrose, sometimes galactose, maltose. Assimilate glucose, sucrose, cellobiose, trehalose, raffinose, melezitose, soluble starch, ethanol, glycerol, erythritol, D-mannitol, D-glucitol, a-methyl-D-glucoside, salicin, DL-Lactate, succinate, citrate, nitrate, vitamin-free (Kurtzman 1998).
Exhibits negative crabtree effect. Produces ethanol under anaerobiosis; acetate under respiratory and respirofermentative growth. Production of ethyl acetate from glucose under oxygen limitation, also other small volatiles, e.g., ethyl propanoate, phenyl ethanol, and 2-phenylethyl acetate. Produces glycerol, arabinitol, and trehalose under osmotic stress and oxygen limitation. High osmotolerance, tolerance to low pH (Passoth et al. 2006).
Ecological Traits:
Fermentation contaminant, soil, grain, ensilage, water, plants (especially fruits and fermenting matter), sewage, warm blooded animals (Kurtzman 1998).
Distinguishing Features: Distinguished from some other strains of Pichia (Hansenula) by high osmotolerance, ferments sucrose, assimilates raffinose, not L-rhamnose (Kurtzman 1998).
Role in wine:
Normal grape flora, active in early fermentation (Renouf et al. 2007). Present in Botrytis-infected fruit (Mills et al. 2002). Can spoil wine by excessive acetic acid and ethyl acetate production (Plata et al. 2003), pellicle formation, and acid metabolism (increasing pH). Effective biocontrol agent of mold on grapes/grapevine (Fleet 2003). Can produce killer factor effective against Dekkera/Brettanomyces spp (Comitini et al. 2004).
Sensitivities:
- SO2: +
- Sorbate: +
- DMDC: +
- pH: –
- Acids: Benzoic
- Ethanol: –
- Anaerobiosis: –
- Heat: –
References:
1. Charoenchai C, Fleet G, Henschke PA. 1998. Effects of temperature, pH and sugar concentration on the growth rates and cell biomass of wine yeasts, American Journal of Enology and Viticulture 49:283–288.
2. Comitini F, Ingeniis De J, Pepe L, Mannazzu I, Maurizio C. 2004. FEMS Microbiology Letters 238:235-240.
3. Fleet G. 2003. Yeast interactions and wine flavour. International Journal of Food Microbiology 86:11-22.
4. Kurtzman CP. 1998. Pichia E.C. Hansen emend. In: Kurtzman CP, Fell JW, eds. The Yeasts, A Taxonomic Study. New York: Elsevier.
5. Mills DA, Johansen EA, Cocolin L. 2002. Yeast diversity and persistence in Botrytis-affected wine fermentations. Applied Environmental Microbiology 68:4884–4893.
6. Passoth V, Fredlund E, Druvors UA, Schnurer J. 2006. Biotechnology, physiology and genetics of the yeast Pichia anomala. FEMS Yeast Research 6:3-13.
7. Plata C, Millan C, Mauricio JC, Ortega JM. 2003. Formation of ethyl acetate and isoamyl acetate by various species of wine yeasts. Food Microbiology 20:217– 224.
8. Renouf V, Claisse O, Lonvaud-Funel A. 2007. Inventory and monitoring of wine microbial consortia. Applied Microbiology and Biotechnology 75:149-164.
9. Warth AD. 1985. Resistance of yeast species to benzoic and sorbic acids and to sulfur dioxide. Journal of Food Protection 48:564–569.
